PARAMOUNT SCHOOL SYSTEM
Subject: Physics – I
Unit 5: Pressure and Deformation in Solids
Exercise
Q.I Choose the best possible option.
-
The most elastic material of the following is:
A. Rubber B. Wood C. Glass D. Steel
-
Hooke’s law holds good up to:
A. proportional limit B. yield limit C. elastic limit D. plastic limit
-
A mass of 2 kg is hung by a spring, which displaces it through 5 cm. The spring constant is:
A. 400 N/m B. 40 N/m C. 4 N/m D. 4000 N/m
-
Materials which do not regain their original shape after removal of the load producing deformation are termed as:
A. Elastic materials B. Plastic materials
C. Rigid materials D. Hooke’s materials
-
SI unit of pressure is:
A. bar B. newton C. psi D. pascal
-
Which will exert greater pressure?
A. 3 g needle of tip area 1 mm²
B. 4000 kg elephant of total feet area 0.5 m²
C. A girl of mass 40 kg wearing high heels of cross-sectional area 0.5 cm²
D. A loaded ship of mass 2.2×107 kg having area 600 m²
-
Pressure of 1000 mbars is equivalent to:
A. 0.1 kPa B. 1 kPa C. 10 kPa D. 100 kPa
-
Pressure of 1 mm Hg is equal to:
A. 1.316×10−3atm B. 1 atm C. 133.29 Pa D. 1.316×105 atm
-
Atmospheric pressure is commonly measured using a:
A. hygrometer B. barometer C. manometer D. thermometer
-
Pressure of liquid in a container increases with:
A. base B. volume C. depth D. mass
-
The atmospheric pressure will be smaller at:
A. Islamabad B. Peshawar C. Lahore D. Murree
-
A girl of mass 50 kg wears heels with an area of 2 cm² in contact with the ground. The pressure she exerts on ground is:
A. 4×10-5Pa B. 4×104 Pa C. 4×10-4Pa D. 4×105Pa
-
Divers wear special suits in order to protect them from:
A. low pressure B. high pressure
C. low temperature D. high temperature
-
In a stationary fluid, the local pressure of the fluid varies:
A. with depth only
B. horizontally only
C. both with depth and along horizontal direction
D. neither with depth nor along horizontal direction
-
The pressure exerted by a man on the surface of the earth will be smaller when he:
A. stands on both feet B. sits on the ground
C. stands on one leg D. sleeps on the ground
SHORT RESPONSE QUESTIONS
QII. Give a short response to the following questions
1. While walking on a trampoline. Do you feel more pressure when you stand still or jump up and down? Why does pressure change with movement?
We experience more pressure when jumping on a trampoline compared to standing still. This happens because, as we jump and land, we exert a greater force due to the combined effect of our body weight and the impact of landing. According to the pressure formula (), pressure increases when force F rises while the contact area A stays the same, causing the trampoline to stretch more under the added impact force.
Pressure changes with movement as the force applied on the same area varies with impact.
2. How does the shape of a thumb pin help it penetrate surfaces easily?
The shape of a thumb pin, with a pointed tip and broad head, helps it penetrate surfaces easily. The pointed tip concentrates force on a very small area, increasing pressure and allowing it to push into surfaces with little effort. The broad head provides an area to apply force comfortably without hurting our fingers.
3. If you blow up a balloon and then tie it closed, why does it stay inflated even though you stop blowing? How does pressure play a role here?
When we blow up a balloon and tie it closed, it stays inflated because of the air pressure inside it. As we blow air into the balloon, we increase the air particles inside, which collide with the balloon’s walls and create internal pressure. This pressure, being greater than the outside air pressure, pushes outward on the balloon’s walls, keeping it inflated even after we stop blowing.
4. Why an inner airtight layer of a space suit is designed to maintain a constant pressure around the astronaut?
An inner airtight layer in a space suit is designed to maintain constant pressure around the astronaut to protect them from the low-pressure environment of space. Without this constant pressure, the body fluids would vaporize due to the lack of external pressure, causing serious harm. The suit’s pressurized layer ensures that astronauts can breathe, move, and function safely in the vacuum of space.
5. If a liquid has density twice the density of mercury, what will be height of liquid column in barometer?
As we know that , pressure depends on both the density of the liquid and the height of the liquid column. If a liquid has a density that is twice that of mercury, the height of its column in a barometer would be half the height of the mercury column for the same atmospheric pressure.
6. Why we wouldn’t be able to sip water with a straw on the moon?
We wouldn’t be able to sip water with a straw on the Moon because of its extremely thin atmosphere, which lacks the necessary pressure to push the liquid up into the straw. On Earth, atmospheric pressure helps lift the liquid when we create a low-pressure area by sucking on the straw, but this principle doesn’t work on the Moon due to its minimal atmospheric pressure and lower gravity.
7. How are we able to break a metal wire by bending it repeatedly?
We can break a metal wire by bending it repeatedly due to the accumulation of fatigue in the material. Each time we bend the wire, tiny cracks may form at the points of stress. Over time, these cracks grow and weaken the wire until it can no longer withstand the applied force, leading to breakage. This process is called metal fatigue, which occurs when a material is subjected to repeated cycles of stress and strain.
8. A spring, having spring constant k when loaded with mass ‘m’, is cut into two equal parts. One of the part is loaded with the same mass m again. What will be its spring constant now?
When a spring is cut into two equal parts, the spring constant of each part becomes double the original spring constant. This is because the spring constant is inversely proportional to the length of the spring.
If the original spring constant is , then the spring constant of each half after cutting will be:
When one of these halves is loaded with the same mass , its new spring constant remains .
9. Why do static fluids always exert a force perpendicular to the surface?
Static fluids exert force perpendicular to a surface because pressure is transmitted equally in all directions, as per Pascal’s Principle. This equal distribution causes fluid particles to push directly outward against surfaces, resulting in a perpendicular force.
10. How can a small car lifter lifts load heavier than itself?
A hydraulic car lifter can lift a load much heavier than it self because of Pascal’s Principle, which states that pressure applied to a fluid is transmitted equally in all directions. In the hydraulic lift, a small force exerted on a piston with a smaller cross-sectional area generates pressure in the hydraulic fluid. This pressure is then transferred to a second, larger piston, which has a larger cross-sectional area. Since the pressure is the same but applied over a larger area, it creates a much larger force, allowing the lifter to raise heavy loads with a smaller input force.
LONG RESPONSE QUESTIONS
QIII. Give a an extended response to the following questions
1. Define elasticity and elastic limit. Show that a force may produce change in size and shape of solids.
Elasticity:
Elasticity is the ability of a deformed body to return to its original shape and size when the deforming forces are removed. For example, when a stretched spring or a distorted ball regains its original shape after the force is no longer applied, this is due to elasticity.
Elastic Limit:
Elastic Limit is the maximum limit up to which a material behaves elastically and can return to its original form. Beyond this limit, the material becomes permanently deformed and cannot return to its original shape or size.
Effects of Force on Solids:
A force applied to a solid can change its size and shape by causing its molecules or atoms to move from their equilibrium positions. When force is applied, like stretching a rubber band or bending a metal rod, the material deforms. When the force is removed, if the material is within its elastic limit, it returns to its original form due to elasticity. However, exceeding the elastic limit causes permanent changes in size and shape.
2. What is Hook’s law? Illustrate its applications. Also, define and calculate spring constant.
Hooke’s Law
Hooke’s Law states that the deformation of a material (such as extension or compression of a spring) is directly proportional to the applied force, provided the material remains within its elastic limit. Mathematically, it is expressed as:
where:
- is the restoring force,
- x is the displacement (extension or compression),
- k is the spring constant, representing the stiffness of the material.
The negative sign indicates that the restoring force is in the opposite direction of displacement.
Applications of Hooke’s Law:
1. Balance Wheel in Mechanical Watches: The balance wheel in a mechanical watch oscillates back and forth, keeping time accurately due to the restoring force provided by the spring’s elasticity.
2. Spring Scale: In a spring scale, Hooke’s Law allows the measurement of weight by observing the extension of the spring when an object is hung from it.
3. Galvanometer: This instrument uses Hooke’s Law to detect electric current by measuring the deflection of a needle, which is restored by a hair spring to its original position.
Spring Constant ():
The spring constant quantifies the stiffness of a spring. It is defined as the ratio of the restoring force to the displacement :
The unit for the spring constant is Newton per meter (N/m).
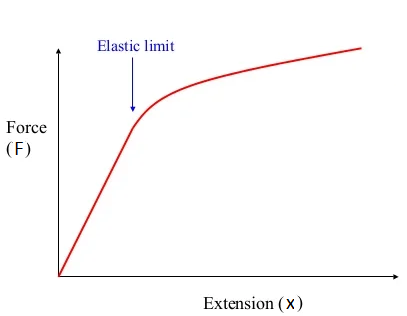
3. Draw and explain force-extension graph for elastic solids.
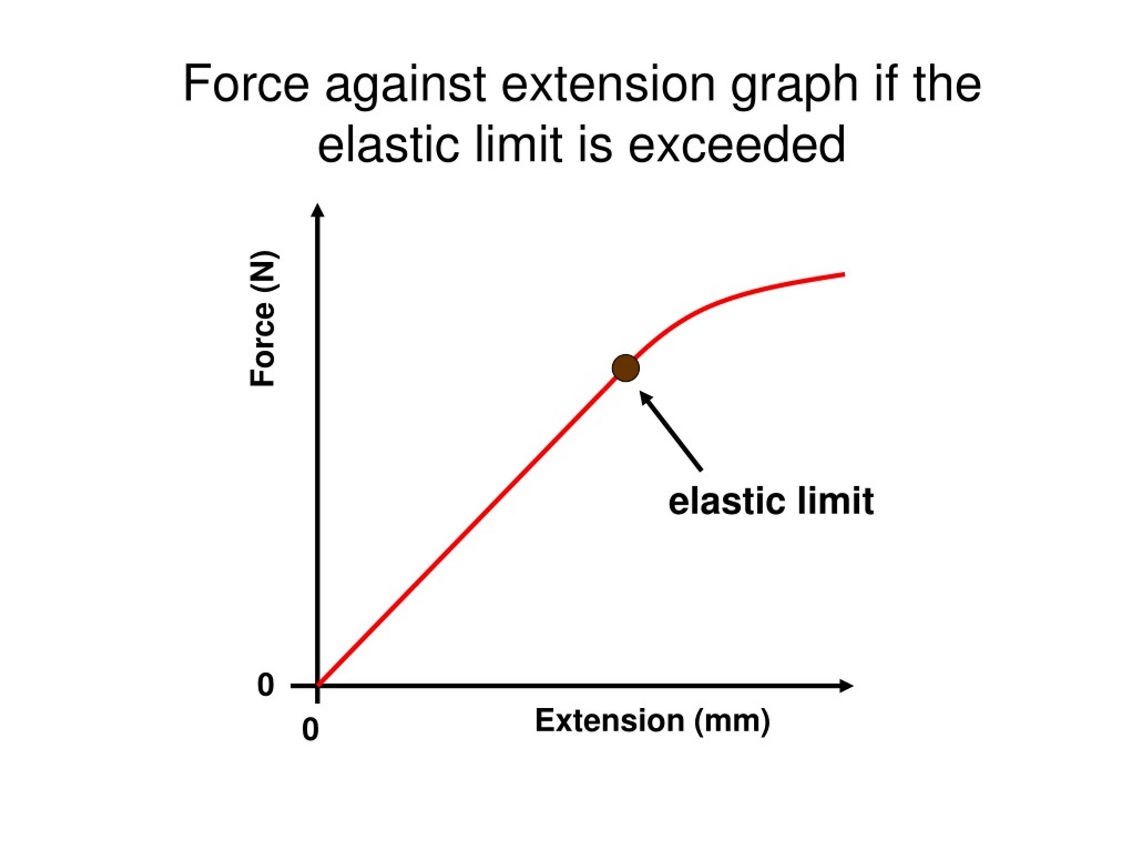
Graph shows a force-extension graph for an elastic solid. There are three parts in this graph.
- Linear Region:
- In the initial portion of the graph, there is a straight line starting from the origin. This indicates that as force is applied, the extension increases proportionally, following Hooke’s Law where. The slope of this linear region represents the stiffness of the material, also known as the spring constant.
- If the force is removed within this region, the solid will return to its original shape, as it is still within its elastic behavior.
- Elastic Limit:
- The point labeled as the elastic limit represents the maximum force the material can withstand while still being able to return to its original shape. Beyond this point, the material no longer obeys Hooke’s Law and begins to deform permanently.
- Up to the elastic limit, the deformation is reversible. After the elastic limit, any further force will cause plastic deformation.
- Nonlinear Region:
- After crossing the elastic limit, the graph curves and no longer shows a straight line. In this region, the extension increases more rapidly for the applied force, indicating plastic deformation.
- Beyond the elastic limit, if the force is removed, the material will not fully return to its original length, indicating permanent deformation.
4. Define and explain pressure. What is effect of area on pressure acting on surface?
Pressure:
Pressure is defined as the force applied per unit area.
- It is represented by the symbol P.
- If a force F is applied to an area A, pressure P is given by:
- The SI unit of pressure is the pascal (Pa), which is equivalent to one newton per square meter (N/m²).
Effect of Area on Pressure:
Pressure is inversely proportional to area. This means that:
- If the area increases, the pressure decreases.
- If the area decreases, the pressure increases.
5. Explain the term atmospheric pressure along with its units. How atmospheric pressure is measured with liquid barometer? Explain its construction and applications.
Atmospheric Pressure:
The pressure that atmospheric particles exert on the surface of earth and all over the surface of objects on the earth is called atmospheric pressure. This pressure acts in all directions and is felt by objects on the Earth’s surface. The average atmospheric pressure at sea level is approximately 1.013×105 N/m2.
Units of Atmospheric Pressure:
- The SI unit of pressure is the pascal (Pa), defined as one newton per square meter (N/m²).
- Another commonly used unit is atmosphere (atm), where 1 atm = 1.013×105 Pa.
- Torr and mmHg are also used, where 1 atm = 760 mmHg = 760 torr.
Liquid Barometer:
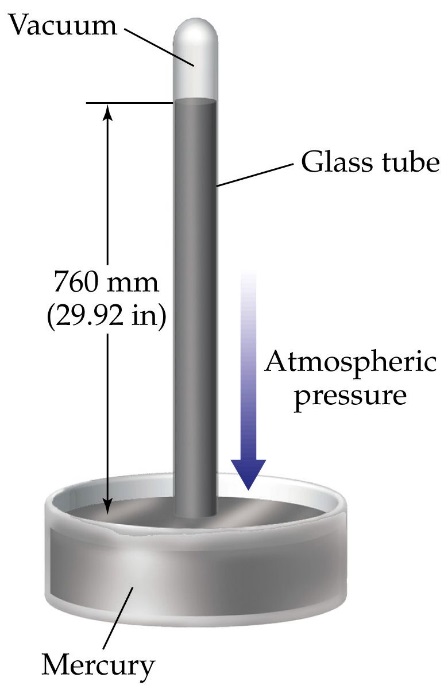
A liquid barometer is a device used to measure atmospheric pressure, most commonly using mercury as the liquid.
Principle of Operation:
A liquid barometer works on the principle of hydrostatic equilibrium, which states that the pressure at a point in a fluid at rest is the same at all depths. Atmospheric pressure balances the weight of the mercury column in the barometer tube.
Construction of a Mercury Barometer:
- A long, closed glass tube is filled with mercury and then inverted into a container also containing mercury.
- As the tube is inverted, mercury flows out until the weight of the mercury column in the tube equals the atmospheric pressure acting on the mercury in the container.
- The height of the mercury column in the tube, typically around 760 mm at sea level, represents the atmospheric pressure.
Applications of a Liquid Barometer:
- Altitude Measurement: As atmospheric pressure decreases with altitude, the barometer’s mercury level drops, allowing for altitude estimation, essential for aviation and setting aircraft altimeters.
- Weather Forecasting: Changes in atmospheric pressure, reflected in the mercury level, help predict weather changes. A falling mercury level indicates potential storms, while a rising level suggests improved weather.
- Industrial Applications: Barometers provide precise pressure readings in industries that require specific atmospheric conditions for processes or equipment operation.
6. Explain with examples how atmospheric pressure varies with altitude. What kind of weather change is indicated by variation in the atmospheric pressure? What are different applications of atmospheric pressure?
Variation of Atmospheric Pressure with Altitude
Atmospheric pressure is the force exerted by air molecules on surfaces. As we ascend from sea level, atmospheric pressure decreases because there are fewer air molecules exerting force. This change is evident in various scenarios:
- At Sea Level: The average atmospheric pressure is about 1 atm or 1.013 × 105 Pa.
- High Altitude (e.g., Mount Everest): At 8.8 km above sea level, the atmospheric pressure is only 33 kPa. This lower pressure makes it harder to breathe as the oxygen levels are reduced due to thinner air.
This decrease in pressure with altitude affects human activities such as mountain climbing and aviation, where special equipment is needed to cope with reduced oxygen availability.
Weather Changes:
Atmospheric pressure is an important indicator of weather patterns:
- Falling Pressure: A decrease in atmospheric pressure often signals an approaching storm or rainy weather, as low pressure areas are associated with moisture-laden air rising and forming clouds.
- Rising Pressure: An increase in atmospheric pressure generally indicates clear or fair weather, as high pressure zones push moisture away, leading to clearer skies.
Applications of Atmospheric Pressure:
- Liquid Barometer for Altitude Measurement: Atmospheric pressure is used in devices like liquid barometers to measure altitude. As pressure decreases with altitude, the mercury column in a barometer drops, allowing pilots to estimate altitude. This is essential in aviation to ensure safe navigation.
- Weather Forecasting: Meteorologists use barometers to track atmospheric pressure changes to predict weather. A falling barometer can indicate an incoming storm, while a rising one suggests improved weather.
- Drinking with a Straw: When we drink through a straw, we reduce the pressure inside our mouth, making it lower than the atmospheric pressure. This pressure difference pushes the liquid up the straw into our mouth.
- Using a Syringe: When the syringe’s plunger is pulled back, the pressure inside decreases. The higher atmospheric pressure outside pushes liquid into the syringe.
- Suction Cups: When air is removed from a suction cup, atmospheric pressure pushes it against a smooth surface, holding it in place.
7. Show that liquid in a container exerts pressure equal to P = pgh. What is effect of depth on pressure of liquid?
Pressure in liquids:
Liquid pressure is the force exerted by a liquid per unit area on the walls of its container or any object submerged in it. This pressure arises due to the weight of the liquid and is directed perpendicular to the surface it acts upon.
Formula:
Derivation:
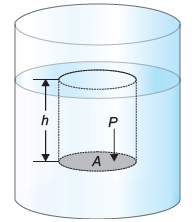
Consider a surface of area “A” in a liquid at a depth “h” as show by shaded region in figure. The length of the cylinder of liquid over this surface will be the weight “w” of the liquid above this surface “p” is the density of the liquid and “m” is mass of liquid above the surface. As we know that;
The mass ” of the cylindrical liquid, in terms of density ‘ ‘ is given by,
Put equation 3 in 2,
……4
Put equation 4 in 1,
Since pressure is defined as,
Now put equation 5 in 6
Dependence:
The above expression shows that liquid pressure depends on three factors:
- Density of liquid (ρ)
- Depth of liquid (h)
Effect of Depth on Liquid Pressure:
The formula shows that liquid pressure increases linearly with depth, meaning greater depth results in higher pressure.
8. State Pascal’s law? Describe working principle of hydraulic lift using Pascal’s law? What do you mean by force multiplier?
Pascal’s Law:
An external pressure applied to an enclosed fluid is transmitted unchanged to every point within the fluid.
Working Principle of a Hydraulic Lift:
A hydraulic lift uses Pascal’s Law. When a force is applied to a small piston with area , it generates a pressure in the hydraulic fluid.
This pressure is transmitted equally to a larger piston with area A2, producing an upward force F2. This allows the lift to raise heavy objects with a small input force.
According to Pascal’s Law,
Putting values,
Force Multiplier:
A force multiplier increases the applied force to achieve a greater output force. In a hydraulic lift, the larger piston area acts as a force multiplier, enabling it to lift heavier loads with minimal effort.
