Chapter Number 2
Q. Write similarities and differences of isotopes?
Ans. Similarities:
Same number of protons.
Same number of electrons
Same atomic numbers.
Differences:
Different number of neutrons.
Different mass numbers.
Q. How does the discovery of isotopes contradicted Dalton’s atomic theory?
Ans. According to Dalton’s atomic theory:
“All atoms of the same element have the same mass”
Whereas isotopes are defined as:
“The atoms of an element that have same atomic number but different mass numbers”.
Which contradicted Dalton’s atomic theory.
Q. Write a note on isotopes of hydrogen?
Ans. Hydrogen has three isotopes.
|
Name |
Symbol |
No. of Proton |
No. of neutrons |
Natural abundance |
|
Protium |
1 |
0 |
99.99% |
|
|
Deuterium |
1 |
1 |
0.0015 |
|
|
Tritium |
1 |
2 |
0% |
Q. Write properties of protium?
Ans. Protium is a colourless, odourless and tasteless gas. It is insoluble in water and is highly inflammable gas.
Q. Write comparison of ordinary water and heavy water?
Ans.
|
Property |
Ordinary water |
Heavy water |
|
Melting point |
0.00°C |
3.81°C |
|
Boiling point |
100°C |
101.2°C |
|
Density at 25°C |
0.99701g/cm3 |
1.1044g/cm3 |
Q. Write a note on isotopes of carbon?
Ans. Carbon has three isotopes.
|
Symbol |
No. of protons |
No. of neutrons |
Natural abundance |
|
6 |
6 |
98.8% |
|
|
6 |
7 |
1.1% |
|
|
6 |
8 |
0.0009% |
Q. Write a note on isotopes of Chlorine?
Ans. There are two isotopes of chlorine.
|
Symbol |
No. of Proton |
No. of Neutrons |
Natural abundance |
|
17 |
18 |
75% |
|
|
17 |
20 |
25% |
Q. What are defects in Rutherford’s atomic model?
Ans. Rutherford’s atomic model has following defects:
- Rutherford suggests that electron being charged particle will emit energy continuously while revolving around the nucleus. Thus the orbit of the revolving electron becomes smaller and smaller until it would fall into the nucleus. This would collapse the atomic structure.
- If revolving electron emits energy continuously it should form a continuous spectrum for an atom but a line spectrum is obtained.
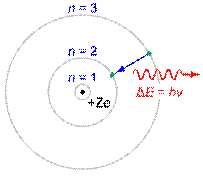
Q. Write postulates of Bohr’s atomic theory?
Ans. In 1913 Neil Bohr, proposed a model for an atom. Main postulates are:-
- The electron in an atom revolves around the nucleus in one of the circular orbits. Each orbit has a fixed energy. So each orbit is also called energy level.
- The energy of the electron in an orbit is proportional to its distance from the nucleus. The farther the electron is from the nucleus, the more energy it has.
- The electron revolves only in those orbits for which the angular momentum of the electron is an integral multiple of where h is Plank’s constant (its value is 6.626×10-34J.s.
- Light is absorbed when an electron jumps to a higher energy orbit and emitted when an electron falls into a lower energy orbit. Electron present in a particular orbit does not radiate energy.
- The energy of the light emitted is exactly equal to the difference between the energies of the orbits.
ΔE=E2-E1
Where ΔE is energy difference between any two orbits with energies E1 and E2
Figure shows Bohr model of the atom
Self-Assessment – Ex 2.1
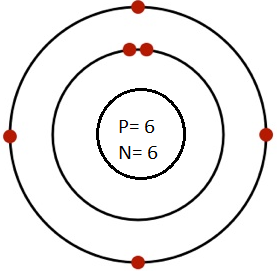
Draw Bohr’s model for the following atoms indicating the location for electrons, protons and neutrons.
(a) Carbon (Atomic No. 6, Mass No. 12)
Ans. No of electron = No. of proton = Atomic No. = 6
Number of neutrons = 12-6=6
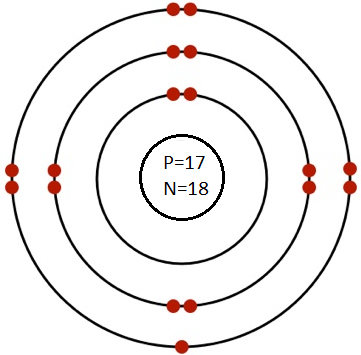
(b) Chlorine (Atomic No. 17, Mass No. 35)
Ans. No. of electron = No. of proton = Atomic No. 17
Number of neutrons = 35-17 = 18
Q. What are isotopes?
Ans. Isotopes is a Greek word “isos” means same and “tope” means place. First used by Soddy. It is defined as :
‘Isotopes are atoms of an element whose nuclei have the same atomic number but different mass number. This is because atoms of an element can differ in the number of neutrons.”
Q. Write a note on isotopes of Uranium?
Ans. There are three isotopes of uranium.
|
Symbol |
No. of Protons |
No. of Neutrons |
Natural abundance |
|
92 |
142 |
0.006% |
|
|
92 |
143 |
0.72% |
|
|
92 |
146 |
99.27% |
Q. Why isotopes of an element have similar chemical properties?
Ans. Chemical properties of an element depend upon the number of protons and electrons. Neutrons do not take part in ordinary chemical reactions. Therefore isotopes of an element have similar chemical properties.
Q. How Carbon-14 is used to estimate the age of carbon containing substances?
Ans. Carbon-14 is used to estimate the age of carbon-containing substances. Carbon atoms circulate between the oceans and living organism at a rate very much faster than they decay. As a result concentration of C-14 increasing. After death organisms no longer pick up C-14. By which we can estimate the date of death. This process is called dating.
Q. What is electronic configuration? Also state Auf Ban principle.
Ans. Electronic configuration:
“The arrangement of electrons in sub-shells is called electronic configuration.”
Auf Ban Principle:
“Electrons fill the lowest energy sub-shell that is available first.”
This means electron will fill first 1s, then 2s, then 2p and so on.
Q. Write electronic configuration of following:
5B
= 1s2 2s2 2p1
6C
= 1s2 2s2 2p2
7N
= 1s2 2s2 2p3
8O
= 1s2 2s2 2p4
9F
= 1s2 2s2 2p5
10Ne
= 1s2 2s2 2p6
11Na
= 1s2 2s2 2p6 3s1
12Mg
= 1s2 2s2 2p6 3s2
Self-Assessment – Ex 2.3
Q. Write the complete electronic configuration for the following elements
Al (atomic number 13)
13Al= 1s2 2s2 2p6 3s2 3p1
Si (atomic number 14)
14Si= 1s2 2s2 2p6 3s2 3p2
P (atomic number 15)
15P= 1s2 2s2 2p6 3s2 3p3
S (atomic number 16)
16S= 1s2 2s2 2p6 3s2 3p4
Cl (atomic number 17)
17Cl= 1s2 2s2 2p6 3s2 3p5
Ar (atomic number 18)
18Ar = 1s2 2s2 2p6 3s2 3p6
Self-Assessment – Ex 2.4
Q. Write the electronic configuration for the following isotopes.
(a)
= 1s2 2s2 2p2
(b)
= 1s2 2s2 2p6 3s2 3p5
(c)
= 1s2 2s2 2p6 3s2 3p5
Review Questions
Q.1 Encircle the correct answer
- According to Bohr atomic model:
- Each orbit has fixed energy, so each orbit is called sub-energy level.
- The energy of the electron is inversely proportional to its distance from the nucleus.
- Light is absorbed when an electron jumps a lower energy orbit.
- The further the electron is from the nucleus, the more energy it has.
- Chlorine has two isotopes. both of which have
- same mass number.
- same number of neutrons.
- different number of protons.
- same number of electrons.
- Number of neutrons in are
- 13
- 14
- 27
- 15
- Which isotope is commonly used to irradiate cancer cells?
- Iodine-123
- Carbon-I4
- Cobalt-60
- Iodine-131
- M-shell has sub-shells:
- 1s,2s
- 2s, 2p
- 3s. 3p, 3d
- 1s, 2s, 3s
- A sub-shell that can accommodate 6 electrons is
- s
- d
- p
- f
- Na has electronic configuration:
- 1s22s23s1
- 1s22s22p7
- 1s22s22p63s2
- 1s22s12p63s1
- Rutherford used ____ particles in his experiments.
- He atoms
- He+
- He2
- He-2
- Which of the following statement is not correct about isotopes?
- they have same atomic number
- they have same number of protons
- they have same chemical properties
- they have same physical properties
- Which isotope is used in nuclear reactors?
- U-234
- U-238
- U-235
- All of these
Answers
|
i. D |
ii. D |
iii. B |
iv. C |
v. C |
|
vi. C |
vii. D |
viii. C |
ix. D |
x. C |
Q.2 Give short answers
(i) Distinguish between shell and sub-shell
Shell:
The electron in an atom revolves around the nucleus in one of the circular paths called shells. Each shall is called energy level. They have given the name as K, L, M, N etc. Each shell have fixed number of electrons, calculated by a formula 2n2.
Sub-shells
Each shell is divided into sub-shells. The number of sub-shells in a shell is equal to its “n” value. Each sub-shell is designated by s,p,d,f etc. The increasing order of energy of the sub-shells is given as:
1s < 2s < 2p < 3s < 3p < 4s < 3d …………
(ii) An atom is electrically neutral, why?
Ans. An atom have the same number of positive protons and negative electrons which cancel each other’s effect and net charge on atom become zero. Hence atom become neutral.
(iii) How many sub-shells are there in N shell?
Ans. N-shell contains four sub-shells, 4s, 4p, 4d, 4f
(iv) Give notation for sub-shells of M shell.
Ans. Notation for sub-shells of M shell are 3s, 3p and 3d.
(v) List the sub shells of M shell in order of increasing energy.
Ans. 3s < 3p < 3d
(vi) Can you identify an atom without knowing number of neutrons in it.
Ans. Yes, we can identify an atom if we know atomic number.
Q.3 The electronic configurations listed are incorrect. Explain what mistakes has been made in each and write correct electronic configurations.
X = 1s2 2s2 2p4 3p2
Y = 1s2 2s1 2p1
Z = 1s2 2s2 2p5 3s1
Ans. Reasons:
According to Aufbau principle, electrons fill the lowest energy sub-shell that is available first.
1s < 2s < 2p < 3s < 3p < 4s < 3d …………
Corrections
X = 1s2 2s2 2p6
Y = 1s2 2s2
Z = 1s2 2s2 2p6
Q.4 Which orbital in each of the following pairs is lower in energy?
a. 2s,2p Ans. 2s < 2p
b. 3p, 2p Ans. 2p < 3p
c. 3s, 4s Ans. 3s < 4s
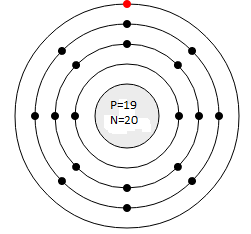
Q.5 Draw Bohr’s Model for the following atoms indicating the location for electron, proton and neutrons
(a) Potassium (Atomic No. 19, Mass No. 39)
(b) Silicon (Atomic No. 14, Mass No. 28)
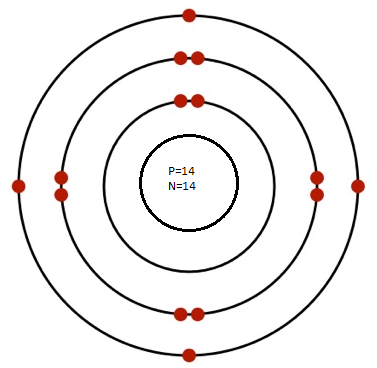
(c) Argon (Atomic No. 18, Mass No. 39)
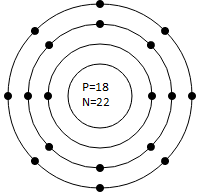
Q.6 Write electronic configuration for the following elements:
(a)
= 1s2 2s2 2p6 3s2 3p2
(b)
= 1s2 2s2 2p6 3s2
(c)
= 1s2 2s2 2p6 3s2 3p1
(d)
= 1s2 2s2 2p6 3s2 3p6
Q.7 Describe the contribution that Ruther ford made to the development of atomic theory.
Ans. Rutherford Atomic Model:
In 1911 Rutherford performed an experiment in order to know the arrangement of electrons and protons in atoms.
Experiment:
Rutherford bombarded α-particles on a 0.0004cm thick gold foil. Alpha particles are emitted by radioactive elements like polonium. These particles are (He++) helium nuclei. Most of these helium nuclei that are doubly positively charged (He++). Most of these particles passed straight through the foil. Only few particles were slightly deflected. But one in 1 million was deflected through an angle greater than 90° from their straight paths. Rutherford performed a series of experiments using this foils of other elements. He observed similar results from these experiments.
Conclusion:
-
-
-
- Since majority of the α-particles passed through the foil undeflected, most of the space occupied by an atom must be empty.
- The deflection of a few α-particles though angles greater than 90º shows that these particles are deflected by electrostatic repulsion between the positively charged a-particles and the positively charged part of atom.
- Massive α-particles are not deflected by electrons.
-
-
On the basis of conclusion, Rutherford proposed model for an atom.
- The electrons revolve around the nucleus, similar to the solar system (planetary model)
- An atom is neutral particle.
- Nucleus is dense positively charged very small region.
Q.8 Explain how Bohr’s atomic theory differed from Rutherford’s atomic theory.
Ans. According to Rutherford atomic model electron being charged particle emit energy continuously and should fall into the nucleus. This would collapse the atomic structure and continuous spectrum is formed for an atom. But Bohr’s suggest that electron moves around the nucleus in a fixed orbit. Each orbit has a fixed energy. Hence line spectrum is formed for an atom.
Summary of differences between two theories
|
Rutherford’s Atomic Theory |
Bohr’s Atomic Theory |
|
|
i. |
It was based upon classical theory |
It was based upon quantum theory |
|
ii. |
Electrons revolve around the nucleus. |
Electrons revolve around the nucleus in orbits of fixed energy |
|
iii. |
No idea about orbits was introduced. |
Orbits had angular momentum |
|
iv. |
Atoms should produce continuous spectrum |
Atom should produce line spectrum |
|
v. |
Atoms should collapse |
Atoms should exist. |
Q.9 Describe the presence of sub shell in a shell
Ans. Each shell is divided into sub-shells. The number of sub shells in a shell is equal to its “n” value. Each sub-shell is designated by s,p,d,f etc.
|
N value |
Shell |
Sub shell |
|
1 |
K |
s, |
|
2 |
L |
s, p |
|
3 |
M |
s, p, d |
|
4 |
N |
s, p, d, f |
Q.10 State the importance and uses of isotopes in various fields of life.
Ans. Importance and uses of isotopes in various fields of life.
- Radioactive iodine-131 is used as tracer in diagnosing thyroid problem.
- Na-24 is used to trace the flow of blood and detect possible constrictions in circulatory system.
- Iodine-123 is used to image the brain.
- Cobalt-60 is used to irradiate cancer cells.
- Carbon-14 is used to trace the path of carbon in photosynthesis.
- Radioactive isotopes are used to determine the molecular structure.
- Radioactive isotopes are used to date rocks, soils, mummies etc.
- Carbon-14 is used to estimate the age of carbon containing substance.
Q.11 The atomic of an element in 23 and its mass number is 56.
(a) How many protons and electrons does an atom of this element have?
Ans. Atomic number = 23
Atomic mass = 56
No. of protons = No. of electrons = Atomic number =23
(b) How many neutrons does this have?
Number of neutrons = Atomic mass – Atomic number
= 56-23 = 38
Q.12 The atomic symbol of aluminum is written as Al. What information do you get from it?
Ans. Atomic number = 13
Atomic mass = 27
No. of protons = No. of electrons = Atomic number=13
Number of neutrons = 27 – 13 = 14
Electronic configuration:
= 1s2 2s2 2p6 3s2 3p1
Period = 3
Group = 2+1 = III A
So aluminum is metal.
Q.13 How testing prevailing theories bring about change in them?
Ans. When some ideas of scientists are not correct, other scientists did not discard their theory. They revised the theory to take into account new discoveries. This shows how testing prevailing theories bring about change in them.
Q.14 How experimental results of some scientists help chemist to formulate new theories and new explanation?
Ans. Scientists performed experiments on the idea of different theories. Sometimes they could not explain some questions using these theories. Chemist leaped over difficulty by using other theories. This show how experimental results of some scientists help chemist to formulate new theories and new explanation.
ThinkTank
- M-24 is a radioactive isotope used to diagnose restricted blood circulation for example in legs. How many electrons protons and neutrons an there in this isotope . Valence shell electronic configuration of M is 3s1.
Ans. Radioactive isotope Na-24 is ,
Number of electrons = 11
Number of protons = 11
Number of neutrons = 24-11 = 13
- Two isotopes of chlorine are and . How do these isotopes differ? How are they alike?
Ans.
|
Differences: |
||
|
Different Number of neutrons |
18 |
20 |
|
Different Mass Number |
35 |
37 |
|
Similarities: |
||
|
Same Number of protons |
17 |
17 |
|
Same number of electrons |
17 |
17 |
|
Same atomic number |
17 |
17 |
- How many electrons can be placed in all of the sub-Shells in the n=2 shell?
Ans. n = 2 is L-shell. Its sub shells are s and p. So, distribution is 2s22p6 and accommodate 8 electrons (by formula 2n2)
- Mass number of an atom indicates total number of protons and neutrons in the nucleus. Can you identify an atom without any neutron?
Ans. Yes, Hydrogen-1 (protium) has no neutron. Its symbol is
- The table shows the nuclei of five different atoms
|
Name of Atom |
Number of Protons |
Number of Neutrons |
|
A |
5 |
6 |
|
B |
6 |
6 |
|
C |
6 |
7 |
|
D |
7 |
7 |
|
E |
8 |
8 |
- Which atom has highest mass number?
Ans. Atom E has the highest mass number.
Mass number = No. of Protons + Number of neutrons
Mass number = 8+8 = 16
- Which two atoms are isotopes?
Ans. Atoms B and C are isotopes.
- Which atom has least number of electrons?
Ans. Atom A has least number of electrons
- Which atom will have electronic configuration 1s22s22p3
Ans. Atom D will have electronic configuration 1s22s22p3
- Which of the atom contains the most number of electrons
Ans. Atom E contains the most number of electron
- Naturally occurring nitrogen has two isotopes N-14 and N-15 which isotope has greater number of electrons?
Ans. N-14 and N-15 have same number of electrons, because isotopes have same number of electrons.
